Research
The research of J.V. is mainly oriented into three fields.
(I) Electrochemistry of bioactive compounds and study their interactions with biomacromolecules as DNA, peptides and proteins, especially membrane proteins, MPs (Scheme 1).
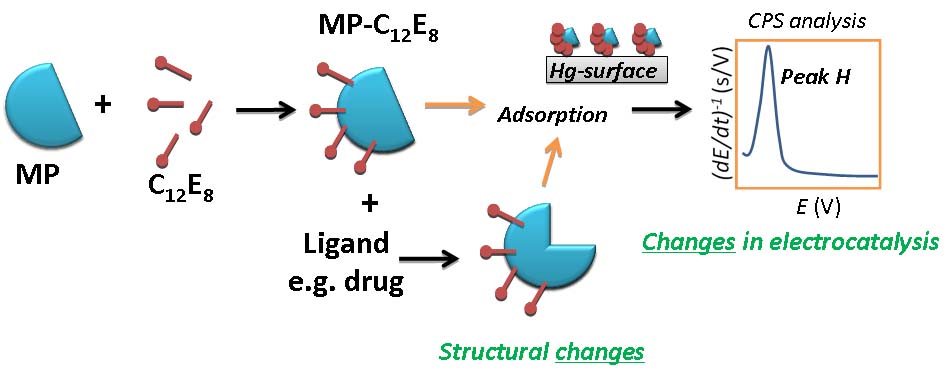
Scheme 1. Simplified representation of the electrocatalytic analysis for the study of molecular interactions and related structural changes of MPs. Abbrev.: C12E8 – non-ionic detergent octaethylene glycol monododecyl ether, MP – membrane protein, CPS – chronopotentiometric stripping. For other details see: Electrochem. Commun., 27, 104–107, 2013; Biochem. Pharmacol., 83, 1507–1513, 2012; Anal. Chem. 87, 1757-1763, 2015; Heterocycles, 88, 879-897, 2014; Anal. Chem. 88, 4548−4556, 2016; Analyst 141, 4554–4557, 2016; Free Radic. Biol. Med. 108, 146−154, 2017
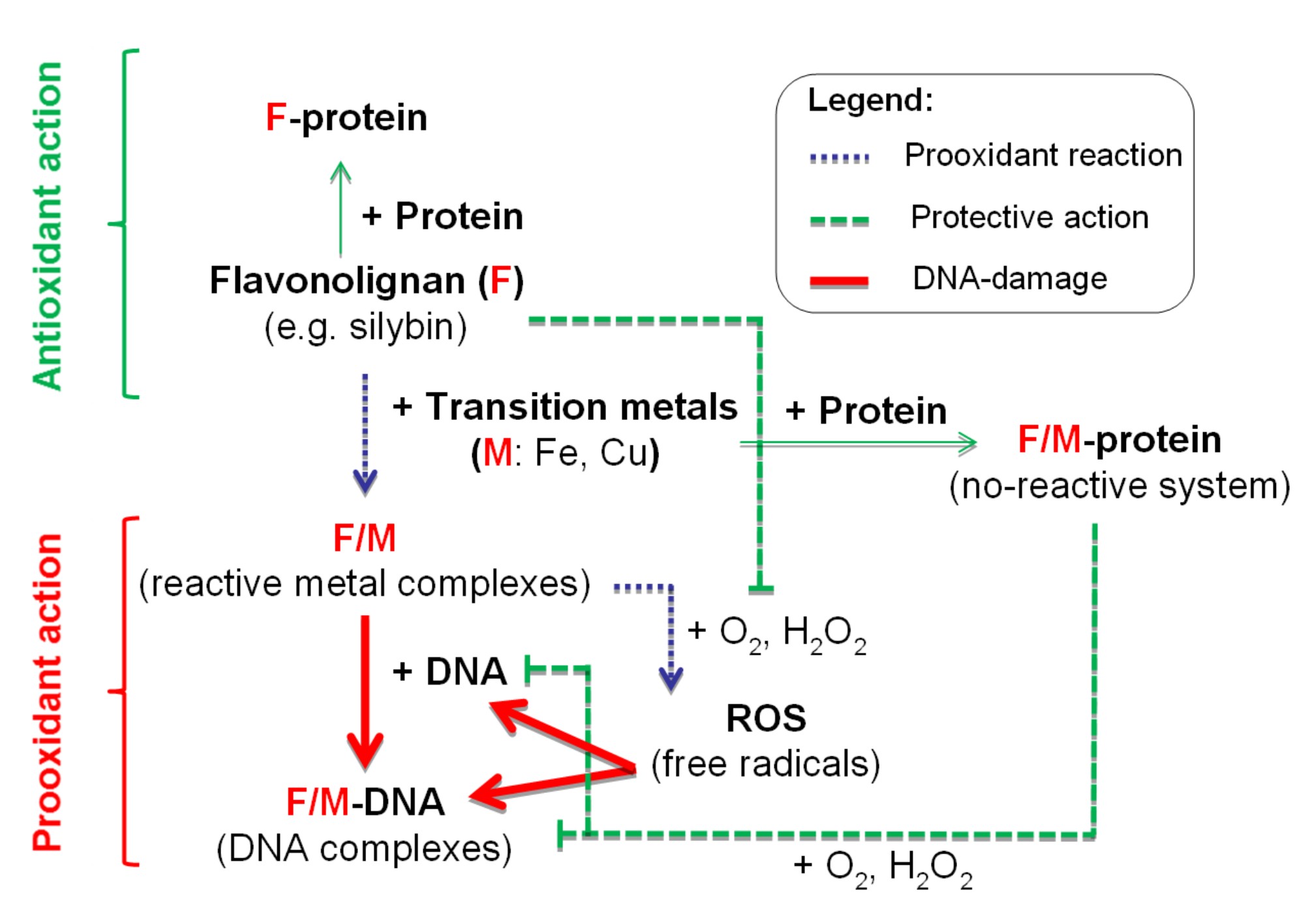
(II) Metabolic studies and mass spectrometry applications in analysis of biotransformation of redox active compounds and drugs. The biotransformations are studied on human hepatocytes in vitro model. For the description of stability, mechanism of oxidative modification and investigation of reactivity of studied compounds, electrochemical tools are frequently used (Scheme 2).
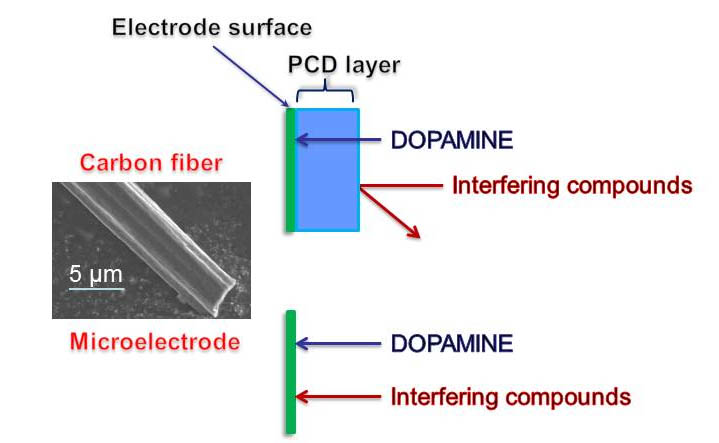
(III) Study of chemical properties and biological activities of cyclopentendiones (CPDs), which is relative new group of bioactive compounds with promising biological and potential pharmacological effects and applications (Scheme 3).
(IV) Preparation of new electrode materials and development of thin conductive layers, chemistry and biochemistry of [n]helicenes and their derivatives (Scheme 4).
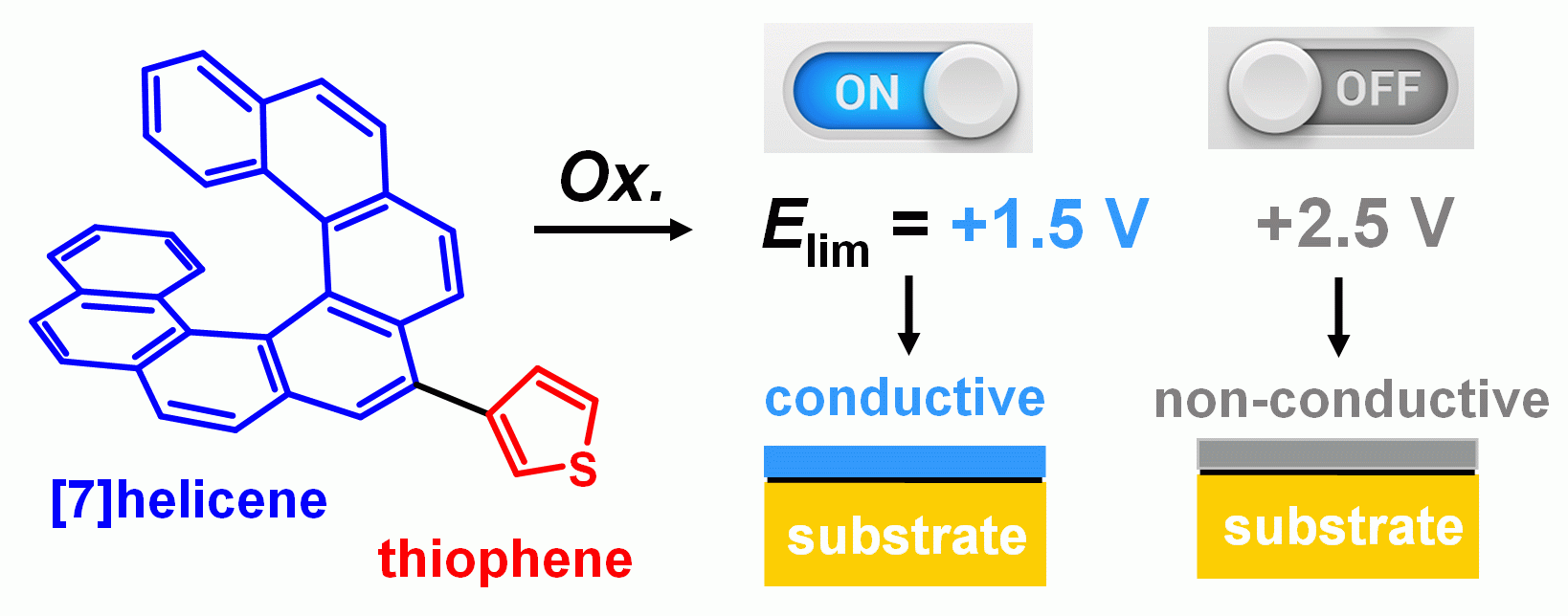
Scheme 4. Polymers based on [7]helicene and thiophene were prepared. Conductive and non-conductive thin layers were synthesized electrochemically. Carbon and indium tin oxide substrates were used for polymer deposition. A joint experimental and computational approach was applied. For other details see: Chem. Eur. J. 21, 2343–2347, 2015; RSC Adv. 4, 46102–46105, 2014; ChemElectroChem 4, 3047–3052, 2017